Information Launch
Tuesday, April 4, 2023

Medication is the very first to minimize bone-weakening system in patients with fibrous dysplasia.

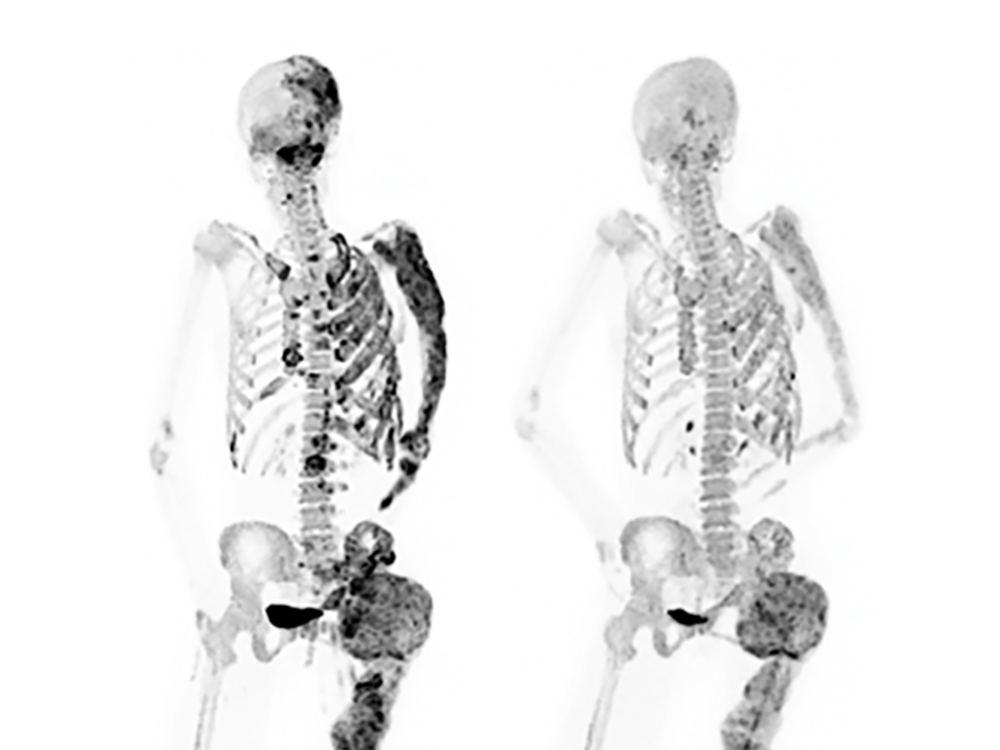
A clinical trial at the Countrywide Institutes of Wellness found that a medication, denosumab, noticeably lessened irregular bone turnover in adults with fibrous dysplasia, a uncommon illness marked by weak and misshapen bones. Bone turnover, a procedure in which previous bone is constantly changed with new bone, is unusually accelerated in fibrous dysplasia and contributes to bone abnormalities. The research of eight individuals was carried out by researchers from the Nationwide Institute of Dental and Craniofacial Exploration (NIDCR) and the NIH Clinical Center. The final results, which confirmed that denosumab might improve patients’ good quality of lifestyle by enabling healthful bone formation, were being revealed as a correspondence report in the New England Journal of Medication.
Fibrous dysplasia stems from gene mutations that lead to scar-like (fibrous) tissue to replace healthful bone setting up in early childhood. These fibrous lesions, which are marked by accelerated bone turnover, weaken bones, primary to bone deformities, fractures, physical disabilities, and agony. In some instances, the lesions can press up from organs and nerves, impairing functions like vision and respiration.
“Surgery is still the normal therapy for fractures and deformities caused by fibrous dysplasia,” said senior author Alison Boyce, M.D., a scientific investigator at NIDCR. “Denosumab is the to start with medication that appears to have an effect on how fibrous dysplasia lesions behave and improves patients’ illness results.”
Denosumab is a medicine licensed by the U.S. Foodstuff and Drug Administration to deal with bone problems from osteoporosis and most cancers. It blocks a protein called RANKL (receptor activator of nuclear component kappa-Β ligand), which is elevated in clients with fibrous dysplasia and is considered to increase bone turnover.
In the stage 2 medical trial, eight ladies been given high doses of denosumab more than six months. At the finish of the cure period, patients’ bone biopsies and scans showed a marked reduction in bone turnover in just lesions. Proteins in the blood connected with bone turnover also dropped to ordinary levels. These effects indicated that the cure improved the good quality and power of patients’ bones.
Sufferers also noted minimized troubles even though getting denosumab. A single affected individual with lesions in the ribs documented greater lung function. Another participant with fibrous dysplasia in the skull expert eyesight advancements.
“This trial is the fruits of 25 many years of scientific investigation at NIDCR to have an understanding of the fundamental mechanisms of fibrous dysplasia and to discover promising treatments,” stated NIDCR Director Rena D’Souza, D.D.S., Ph.D.
Although denosumab showed promise, abnormal bone turnover returned for all but one particular of the sufferers soon after they stopped taking the treatment. In four patients, bone turnover exceeded pre-treatment stages. Dr. Boyce noted that it will be critical for clinicians to issue in these challenges when managing sufferers.
To determine no matter whether earlier intervention with denosumab can prevent lesions from forming entirely, Dr. Boyce and her group are now testing the medicine in a new medical demo for little ones with fibrous dysplasia.
This analysis was supported by the intramural study plan of the Countrywide Institute of Dental and Craniofacial Study (NIDCR) and the NIH Scientific Middle.
About the Countrywide Institute of Dental and Craniofacial Analysis: NIDCR is the nation’s primary funder of investigation on oral, dental, and craniofacial health and fitness.
About the National Institutes of Well being (NIH):
NIH, the nation’s healthcare investigate agency, incorporates 27 Institutes and Facilities and is a ingredient of the U.S. Section of Well being and Human Expert services. NIH is the principal federal company conducting and supporting basic, scientific, and translational health-related research, and is investigating the leads to, remedies, and cures for both typical and scarce health conditions. For more information about NIH and its packages, check out www.nih.gov.
NIH…Turning Discovery Into Health®
References
de Castro LF, Michel Z, Pan K, Taylor J, Szymczuk V, Paravastu S, Saboury B, Papadakis GZ, Li X, Milligan K, Boyce B, Paul SM, Collins MT, Boyce AM. Security and Efficacy of Denosumab for Fibrous Dysplasia of Bone. N Engl J Med. 2023 Feb 23388(8):766-768. DOI: 10.1056/NEJMc2214862




More Stories
White House plans to nominate cancer center chief to lead NIH
What Happens to the Brain in Alzheimer’s Disease?
Biden Plans to Pick Monica Bertagnolli to Lead National Institutes of Health